![joliére, joliere, HA Filler, CE approved, Dermal Filler, CE certified HA Filler [HA Filler]](https://www.beautetrade.com/uploads/images/products/3/2/joliere-joliere-ha-filler-ce-approved-dermal-filler-ce-certified-ha-filler-ha-filler1-0333459001774952039.jpg)
![joliére, joliere, HA Filler, CE approved, Dermal Filler, CE certified HA Filler [HA Filler]](https://www.beautetrade.com/uploads/images/products/0/6/joliere-joliere-ha-filler-ce-approved-dermal-filler-ce-certified-ha-filler-ha-filler2-0611107001774952039.jpg)
![joliére, joliere, HA Filler, CE approved, Dermal Filler, CE certified HA Filler [HA Filler]](https://www.beautetrade.com/uploads/images/products/4/6/joliere-joliere-ha-filler-ce-approved-dermal-filler-ce-certified-ha-filler-ha-filler3-0053815001774952040.jpg)
![joliére, joliere, HA Filler, CE approved, Dermal Filler, CE certified HA Filler [HA Filler]](https://www.beautetrade.com/uploads/images/products/8/1/joliere-joliere-ha-filler-ce-approved-dermal-filler-ce-certified-ha-filler-ha-filler4-0333915001774952040.jpg)
| Size | 71 * 26 * 193 mm |
| Weight | 50 g |
| Stock | 10,000 Boxes |
| Country of Origin | South Korea |
| Production method | Available, OBM, ODM, OEM |
| Certification | CE |
| Ingredients | HYALURONANIC ACID + HYALURONANIC ACID - |
| Hs Code | 3304999000 |



• The jolière is made using ‘HCCL™ technology’ including minimized BDDE, low temperature & long time cross-linkingreaction and a patent process.
• A stabilized 3D matrix structure for high safety and optimal rheological properties.
• The safety and efficacy of jolière is proven through the comparative clinical trial.
joliére
◀ REWIND YOUR TIME
Cross-linked HA Dermal Filler, CE Appreved![]()
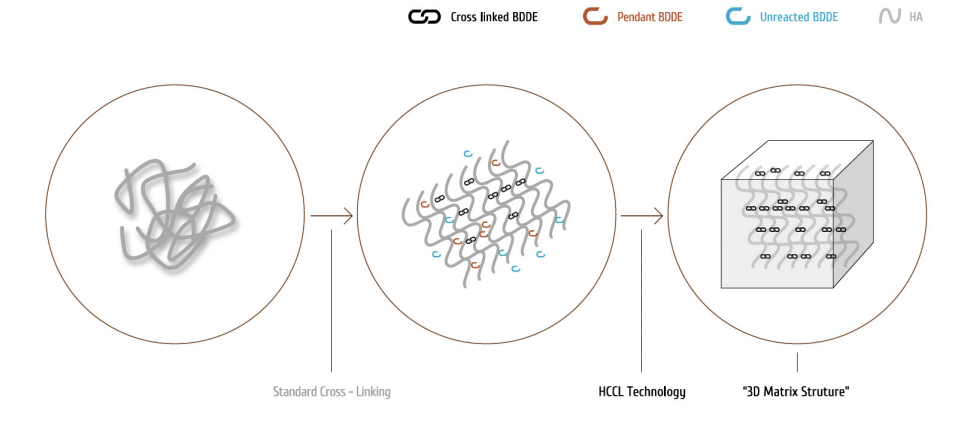
◀ HCCL™ technology(Highly Completely Cross-Linking)
The jolière is made using ‘HCCL™ technology’ including minimized BDDE, low temperature & long time cross-linkingreaction and a patent process.(Patent No. 10-1223652)
 >
>
The jolière is designed to provide the ideal balanced level of cohesiveity, elasticity and plasticity for purpose through
the HCCL™ tcechnology.
◀HCCL™ Technology
A stabilized 3D matrix structure for high safety and optimal rheological properties.
◀Proved Safety
The safety and efficacy of jolière is proven through the comparative clinical trial.
◀CE Mark, ISO 13485
The jolière is CE certified, Which means the product complies with the EU health,
safety and environmental regulations for medical devices Class III.
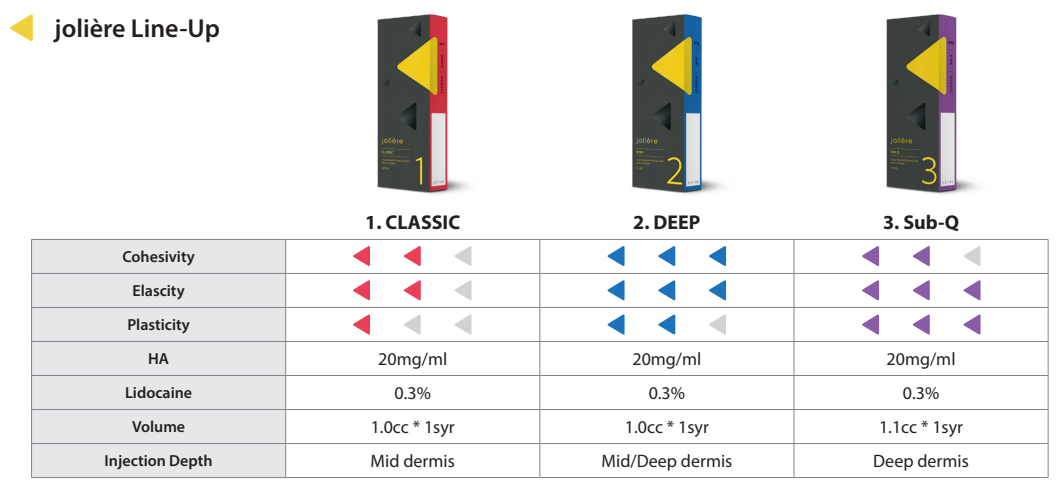
◀Specification
 >
>
 South Korea
South Korea 6F, z152, 602, Yeongdong-daero, Gangnam-gu, Seoul, Republic of Korea
Contact Person Kevin Kim